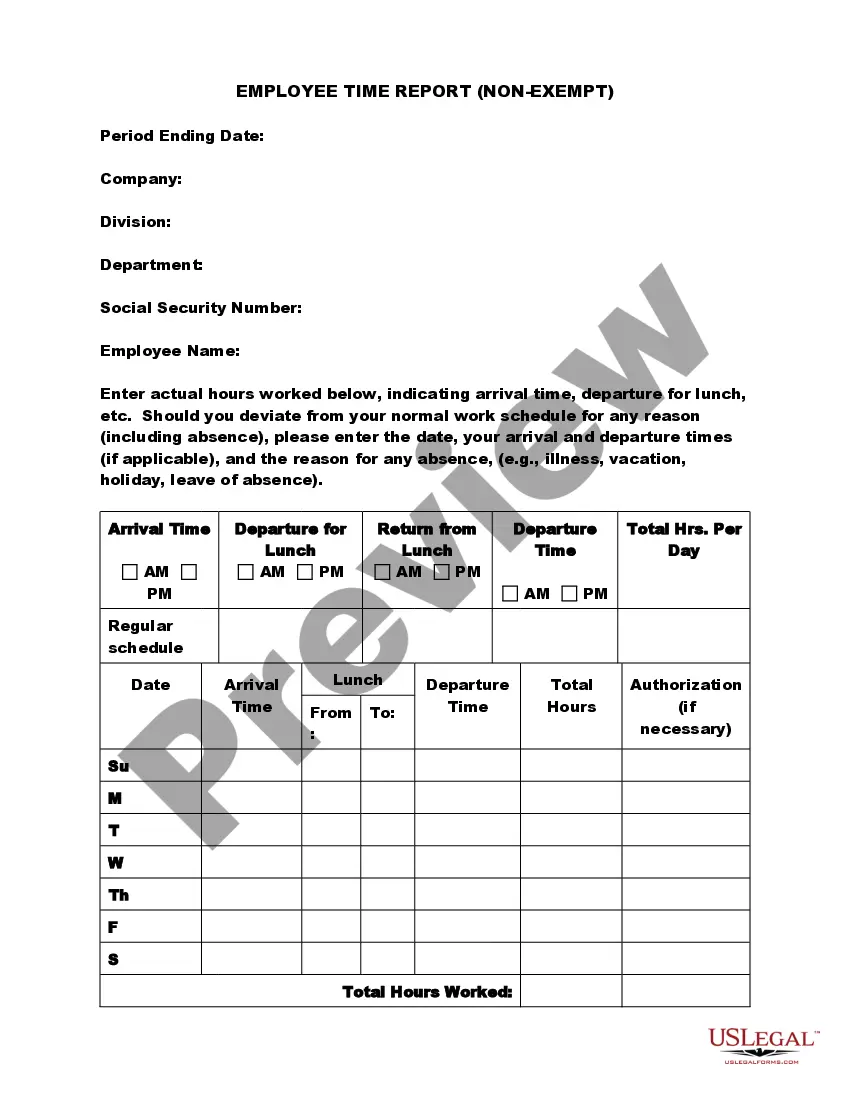
Michigan Exempt Survey
Description
Get your form ready online
Our built-in tools help you complete, sign, share, and store your documents in one place.
Make edits, fill in missing information, and update formatting in US Legal Forms—just like you would in MS Word.
Download a copy, print it, send it by email, or mail it via USPS—whatever works best for your next step.
Sign and collect signatures with our SignNow integration. Send to multiple recipients, set reminders, and more. Go Premium to unlock E-Sign.
If this form requires notarization, complete it online through a secure video call—no need to meet a notary in person or wait for an appointment.
We protect your documents and personal data by following strict security and privacy standards.
Make edits, fill in missing information, and update formatting in US Legal Forms—just like you would in MS Word.

Download a copy, print it, send it by email, or mail it via USPS—whatever works best for your next step.

Sign and collect signatures with our SignNow integration. Send to multiple recipients, set reminders, and more. Go Premium to unlock E-Sign.
If this form requires notarization, complete it online through a secure video call—no need to meet a notary in person or wait for an appointment.
We protect your documents and personal data by following strict security and privacy standards.
Looking for another form?
How to fill out Exempt Survey?
Locating the appropriate legal document template may pose a challenge. Clearly, there is a multitude of designs available online, but how can you identify the legal form you require? Utilize the US Legal Forms website.
This service provides thousands of templates, including the Michigan Exempt Survey, which you can utilize for both business and personal purposes. All forms are vetted by experts and comply with federal and state regulations.
If you are already registered, Log In to your account and click the Download option to locate the Michigan Exempt Survey. Use your account to review the legal documents you have previously ordered. Navigate to the My documents tab in your account and obtain another copy of the document you require.
Complete, modify, print, and sign the acquired Michigan Exempt Survey. US Legal Forms is the largest repository of legal documents where you can discover a wide range of document templates. Use this service to obtain professionally crafted documents that adhere to state requirements.
- If you are a new user of US Legal Forms, here are straightforward steps to follow.
- First, ensure you have selected the correct form for your city/county. You can preview the form using the Review option and examine the form summary to confirm it is suitable for you.
- If the form does not meet your needs, utilize the Search feature to find the appropriate form.
- Once you are confident that the form is adequate, click on the Purchase now option to obtain the form.
- Choose the pricing plan you desire and fill in the required information. Create your account and complete your purchase with your PayPal account or credit card.
- Select the document format and download the legal template for your use.
Form popularity
FAQ
To qualify as an exempt study, the research must fall within one of the specific federal regulatory categories. A determination of exemption must be made by the JHM IRBs and principal investigators must submit all such studies for their review.
Research involving children can be classified as exempt under this category if the research involves only educational tests and / or observation of public behavior where the investigator does not participate in the activities being observed and meets the other conditions of the exempt category.
Exempt research are human subjects studies that present no greater than minimal risk to subjects and fit into one or more exempt categories (as described below).
According to the Federal regulations (45 CFR 46.101(b)), survey research may be exempt from the regulations unless "the information obtained is recorded in such a manner that the human subjects can be identified, directly or through identifiers linked to the subjects" or if "federal statute(s) require(s) without
Exempt Categories:Education research.Surveys, interviews, educational tests, public observations (that do not involve children)Benign behavioral interventions.Analysis of previously-collected, identifiable info/specimens.Federal research/demonstration projects.Taste and food evaluation studies.
Human subjects research that is classified as exempt means that the research qualifies as no risk or minimal risk to subjects and is exempt from most of the requirements of the Federal Policy for the Protection of Human Subjects, but is still considered research requiring an IRB review for an exemption determination.
Exempt human subjects research is a sub-set of research involving human subjects that does not require comprehensive IRB review and approval because the only research activity involving the human subjects falls into one or more specific exemption categories as defined by the Common Rule.
Human subjects research studies that do not qualify for an exemption are referred to as non- exempt human subjects research. Unless there is a Secretarial waiver, they must comply with the Common Rule requirements, including IRB review and approval, before the research can begin.
Human subjects research that is classified as exempt means that the research qualifies as no risk or minimal risk to subjects and is exempt from most of the requirements of the Federal Policy for the Protection of Human Subjects, but is still considered research requiring an IRB review for an exemption determination.
Educational tests, surveys, interviews, or observations research involving the use of educational tests (cognitive, diagnostic, aptitude, achievement), survey procedures, interview procedures, or observations of public behavior meets the definition of human subjects research and requires IRB review.