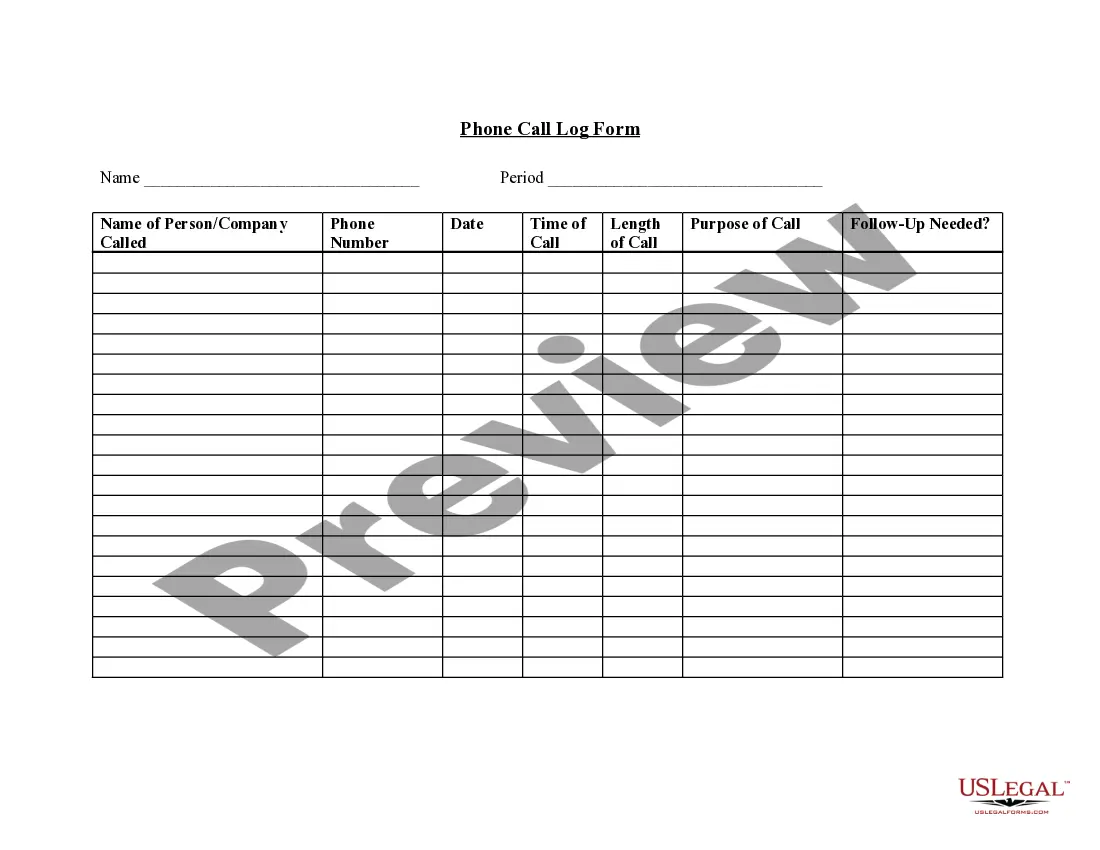
Consent Form For Release Of Information In California
Description
Form popularity
FAQ
I participant name, agree to participate or agree to participation of my child participant name in the research project titled project title, conducted by researcher(s) name who has (have) discussed the research project with me. I have received, read and kept a copy of the information letter/plain language statement.
I participant name, agree to participate or agree to participation of my child participant name in the research project titled project title, conducted by researcher(s) name who has (have) discussed the research project with me. I have received, read and kept a copy of the information letter/plain language statement.
(b) Informed consent must include a verbal explanation by a physician of the client's right to refuse or accept medical treatment. It must include a written consent form signed by the client indicating the above information has been given.
How to write a consent form: A step-by-step guide Step 1: Title and introduction. Step 2: Description of the activity. Step 3: Risks and benefits. Step 4: Confidentiality and data handling. Step 5: Voluntary participation and withdrawal. Step 6: Consent statement. Step 7: Signature and date. Step 8: Contact information.
It must be obvious that the individual has consented, and what they have consented to. This requires more than just a confirmation that they have read terms and conditions – there must be a clear signal that they agree. If there is any room for doubt, it is not valid consent.
A document with important information about a medical procedure or treatment, a clinical trial, or genetic testing. It also includes information on possible risks and benefits. If a person chooses to take part in the treatment, procedure, trial, or testing, he or she signs the form to give official consent.
Instructions for Developing an Informed Consent Document General Information. Describe the purpose(s) of this research study in lay terms. Purpose of the Study. Procedures. Risks. Benefits. Compensation, Costs and Reimbursement. Withdrawal or Termination from Study. Confidentiality.
Examples of giving nonverbal consent may include: Head nod. Thumbs up. Pulling someone closer. Nodding yes. Making direct eye contact. Actively touching someone. Initiating sexual activity.
Generally, an authorization provides the authority for a doctor's release of PHI for specified purposes, which are generally other than treatment, payment, or healthcare operations, or, to disclose protected health information to a third party specified by the individual.
Content for a valid authorization includes: The name of the person or entity authorized to make the request (usually the patient) The complete name of the person or entity to receive the protected health information (PHI) A specific description of the information to be used or disclosed, including the dates of service.