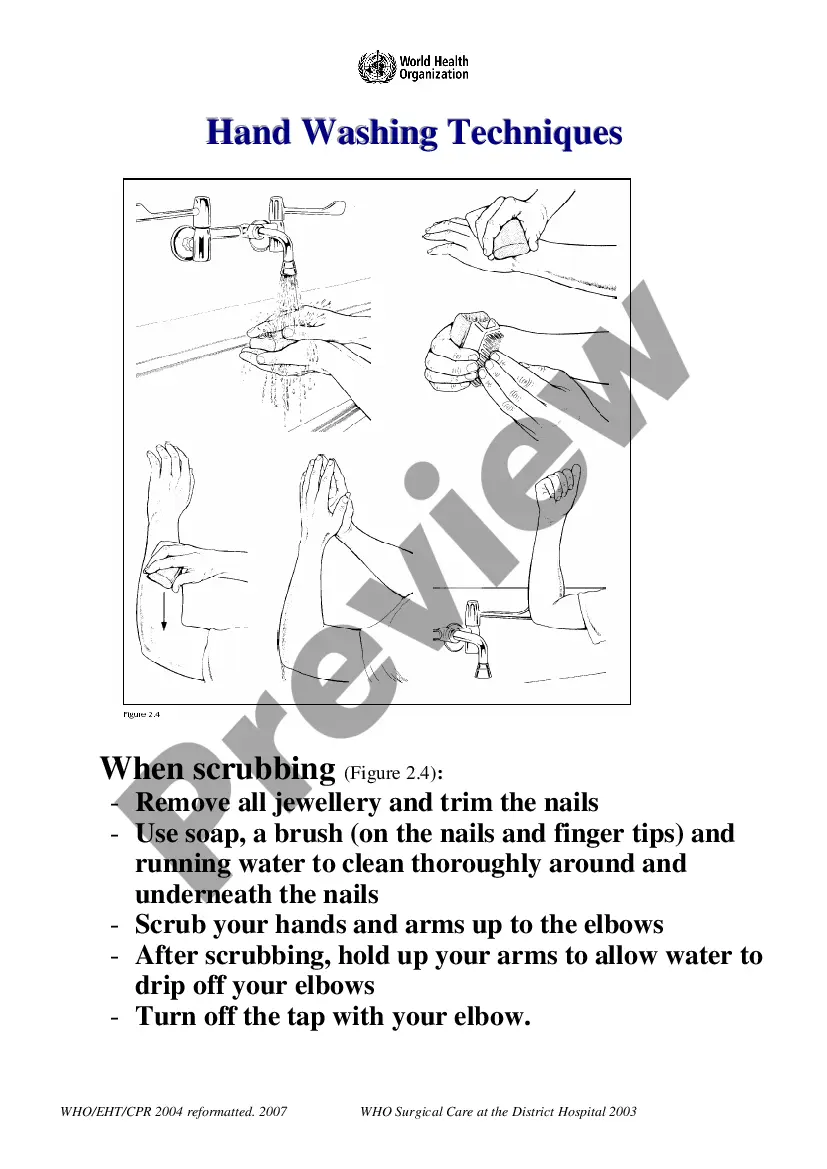
Best Practice Protocols Clinical Procedures Safety
Does this form need to be notarized?
This form does not typically require notarization to be legally valid. However, some jurisdictions or document types may still require it. US Legal Forms provides secure online notarization powered by Notarize, available 24/7 for added convenience.
Get your form ready online
Our built-in tools help you complete, sign, share, and store your documents in one place.
Make edits, fill in missing information, and update formatting in US Legal Forms—just like you would in MS Word.
Download a copy, print it, send it by email, or mail it via USPS—whatever works best for your next step.
Sign and collect signatures with our SignNow integration. Send to multiple recipients, set reminders, and more. Go Premium to unlock E-Sign.
If this form requires notarization, complete it online through a secure video call—no need to meet a notary in person or wait for an appointment.
We protect your documents and personal data by following strict security and privacy standards.
Make edits, fill in missing information, and update formatting in US Legal Forms—just like you would in MS Word.

Download a copy, print it, send it by email, or mail it via USPS—whatever works best for your next step.

Sign and collect signatures with our SignNow integration. Send to multiple recipients, set reminders, and more. Go Premium to unlock E-Sign.
If this form requires notarization, complete it online through a secure video call—no need to meet a notary in person or wait for an appointment.
We protect your documents and personal data by following strict security and privacy standards.
Looking for another form?
Form popularity
FAQ
Our clinical trials registry is available online at www.clinicaltrials.gov. Additionally, clinical trial protocol information submitted by AbbVie to the EudraCT database is made publicly available by the EMA via the clinical trials register.
What is a Nursing Protocol? 25a1 A nursing protocol is considered to be a set of. predetermined criteria that define appropriate nursing interventions that articulate or describe situations in which the nurse makes judgments relative to a course of action for effective management of common patient care problems.
The protocol is a document that describes how a clinical trial will be conducted (the objective(s), design, methodology, statistical considerations and organization of a clinical trial,) and ensures the safety of the trial subjects and integrity of the data collected.
A protocol can be described as an agreement to a particular sequence of activities that assist health care workers to respond consistently in complex areas of clinical practice. Protocols may be established on a unidisciplinary or multidisciplinary basis.
However, the use of checklists and protocols clearly has been demonstrated to improve outcomes and their use is strongly encouraged. Checklists and protocols should be incorporated into systems as a way to help practitioners provide the best evidence-based care to their patients.
Clinical protocols are basically rules of how to proceed in certain situations. They provide health care practitioners with parameters in which to operate. The term 'code of practice' may be used synonymously with clinical protocols. A code comprises a set of laws or rules.
The protocol should outline the rationale for the study, its objective, the methodology used and how the data will be managed and analysed. It should highlight how ethical issues have been considered, and, where appropriate, how gender issues are being addressed.
The use of clinical protocols allows health care providers to offer appropriate diagnostic treatment and care services to patients, variance reports to purchasers and quality training to clinical staff.
Nursing protocols in high-acuity and critical care settings help prevent complications, promote faster recovery, enhance patient safety, increase nursing autonomy and reduce costs, according to expert critical care nurses such as Ramon Lavandero, RN, MA, MSN, FAAN, director, communications and strategic alliances for