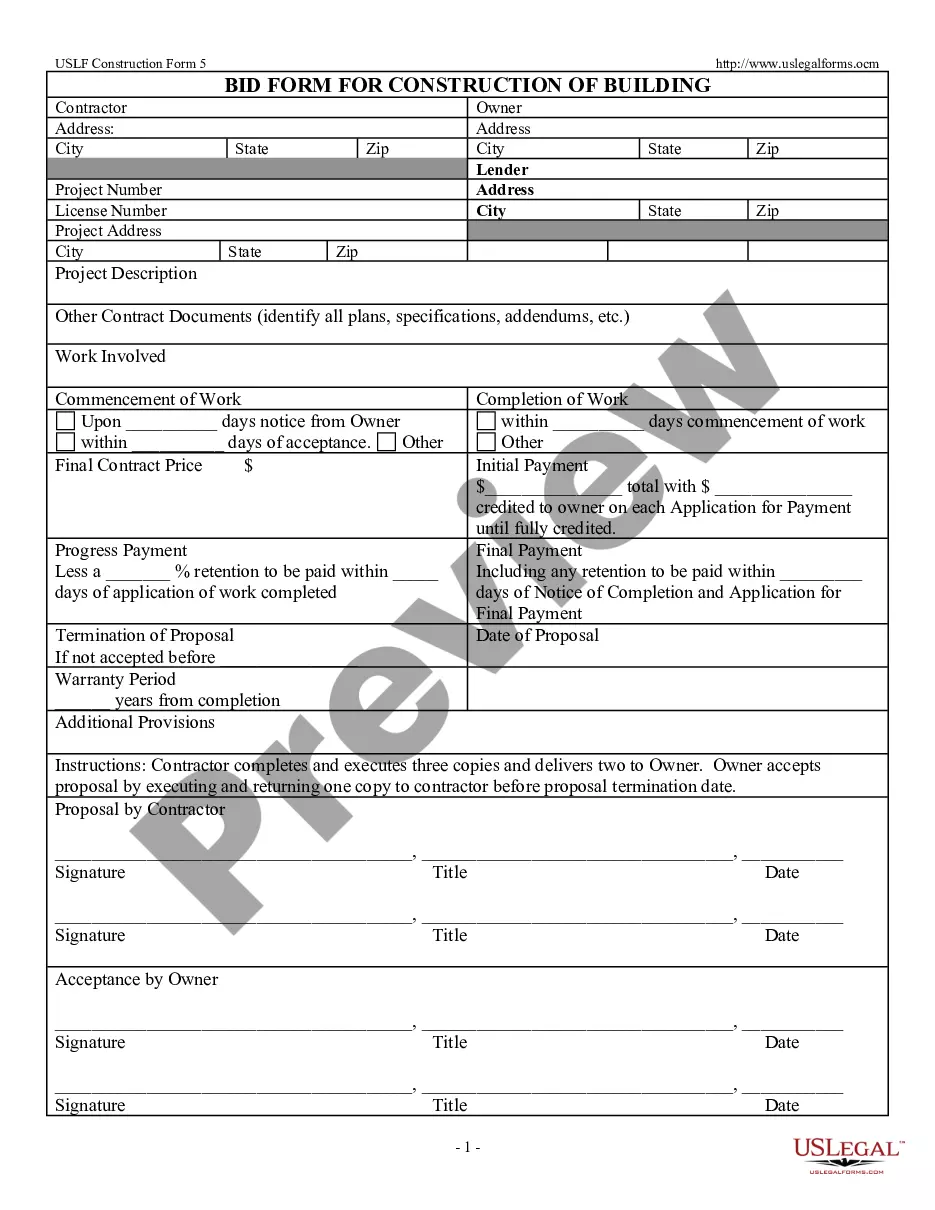
Consent Release Information Form Psychologist In Clark
Description
Form popularity
FAQ
Instructions for Developing an Informed Consent Document General Information. Describe the purpose(s) of this research study in lay terms. Purpose of the Study. Procedures. Risks. Benefits. Compensation, Costs and Reimbursement. Withdrawal or Termination from Study. Confidentiality.
“Informed Consent, psychologists inform participants about (1) the purpose of the research, expected duration and procedures; (2) their right to decline to participate and to withdraw from the research once participation has begun; (3) the foreseeable consequences of declining or withdrawing; (4) reasonably foreseeable ...
At a minimum, a well-designed informed consent form will address the following information: Risks and benefits of treatment. Fees and payment policies. Confidentiality and its limits. Contact information and communication. Social media policy and general boundaries. Emergency procedures.
I understand the information provided for the study insert title as described herein. My questions have been answered to my satisfaction, and I agree to participate in this study. I have been given a copy of this form.
The entire informed consent process involves giving a subject adequate information concerning the study, providing adequate opportunity for the subject to consider all options, responding to the subject's questions, ensuring that the subject has comprehended this information, obtaining the subject's voluntary agreement ...
Instructions for Developing an Informed Consent Document General Information. Describe the purpose(s) of this research study in lay terms. Purpose of the Study. Procedures. Risks. Benefits. Compensation, Costs and Reimbursement. Withdrawal or Termination from Study. Confidentiality.
The five essential elements of the informed consent process are Voluntary, Information, Understanding, Capacity, and Consent. Voluntary: Informed consent should be given voluntarily without any coercion or pressure.
Obtaining informed consent in medicine is a process that should include describing the proposed intervention, emphasizing the patient's role in decision-making, discussing alternatives to the proposed intervention, discussing the risks and benefits of the proposed intervention, and eliciting the patient's preference, ...
It involves understanding the person's goals and concerns, and discussing with the person (or their substitute decision-maker) their options for treatment, the potential outcomes (positive, negative and neutral), risks and benefits and what this might mean for them.
Consent should be obtained before the participant enters the research (prospectively), and there must be no undue influence on participants to consent. The minimum requirements for consent to be informed are that the participant understands what the research is and what they are consenting to.