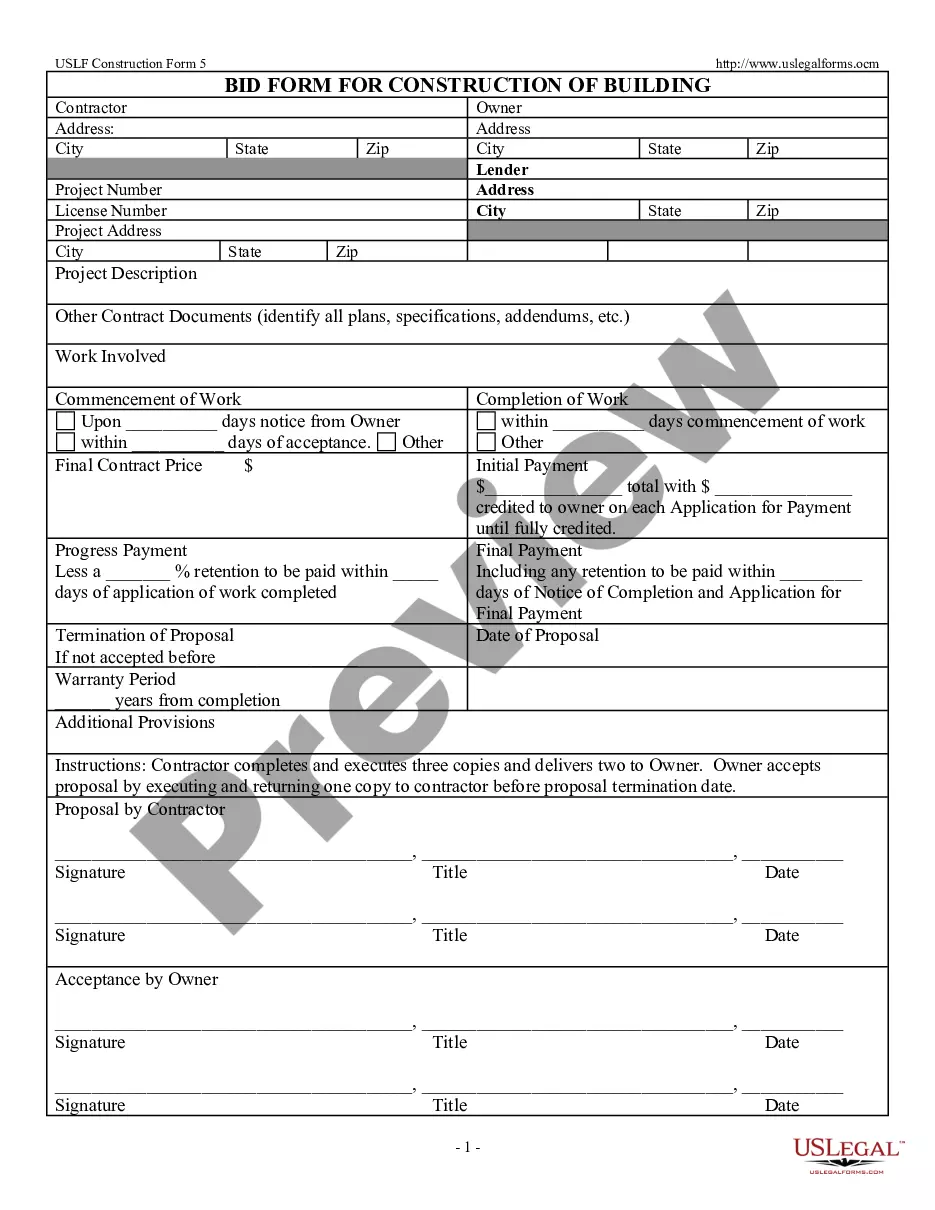
Trial Would Attorney Withdraw In Montgomery
Description
Form popularity
FAQ
38% of patients who dropped out of a study early thought the site visits were stressful compared to 16% who completed the trial. 47% of patients who dropped out of a study early said they were motivated by “myself” to stay enrolled in the study compared to 78% who completed the trial.
You may file a formal complaint against an attorney with the Office of Disciplinary Counsel (ODC), which is the division of the Disciplinary Board of the Supreme Court of Pennsylvania that is charged with investigating complaints against attorneys for violations of the Rules of Professional Conduct (RPC).
FDA recognizes that a subject may withdraw from a study; however, the withdrawal does not extend to the data already obtained during the time the subject was enrolled.
When withdrawing from a study, let the research team know that you want to withdraw. The research team may ask why you want to leave the study. It can be helpful to have this information but you do not have to provide a reason if it makes you uncomfortable.
• If a participant no longer wishes to. remain in a study, that is considered. withdrawal of consent.
A participant can leave a research study at any time. When withdrawing from the study, the participant should let the research team know that they wish to withdraw. A participant may provide the research team with the reason(s) for leaving the study, but is not required to provide their reason.
Sometimes the attorney is the one who feels that the client and the lawyer should no longer work together. When this happens, the attorney “withdraws” from the case and terminates the relationship and ceases to represent the client.
Call the Disciplinary Board of the Supreme Court of Pennsylvania at 717-231-3380 or go to their website.
Notice of Intent to Withdraw means a City approved form giving notice of an Owner's intent to withdraw a building containing at least one Covered Unit from the residential rental market in ance with Government Code sections 7060 – 7060.7.
PA Office of Attorney General (OAG) investigates trade practices in the distribution, financing and furnishing of goods and services for possible unfair and deceptive conduct in PA. Submit a complaint online or call (717) 787-9707 or (800) 441-2555.