Confidentiality Disclosure Agreement In Clinical Research In Middlesex
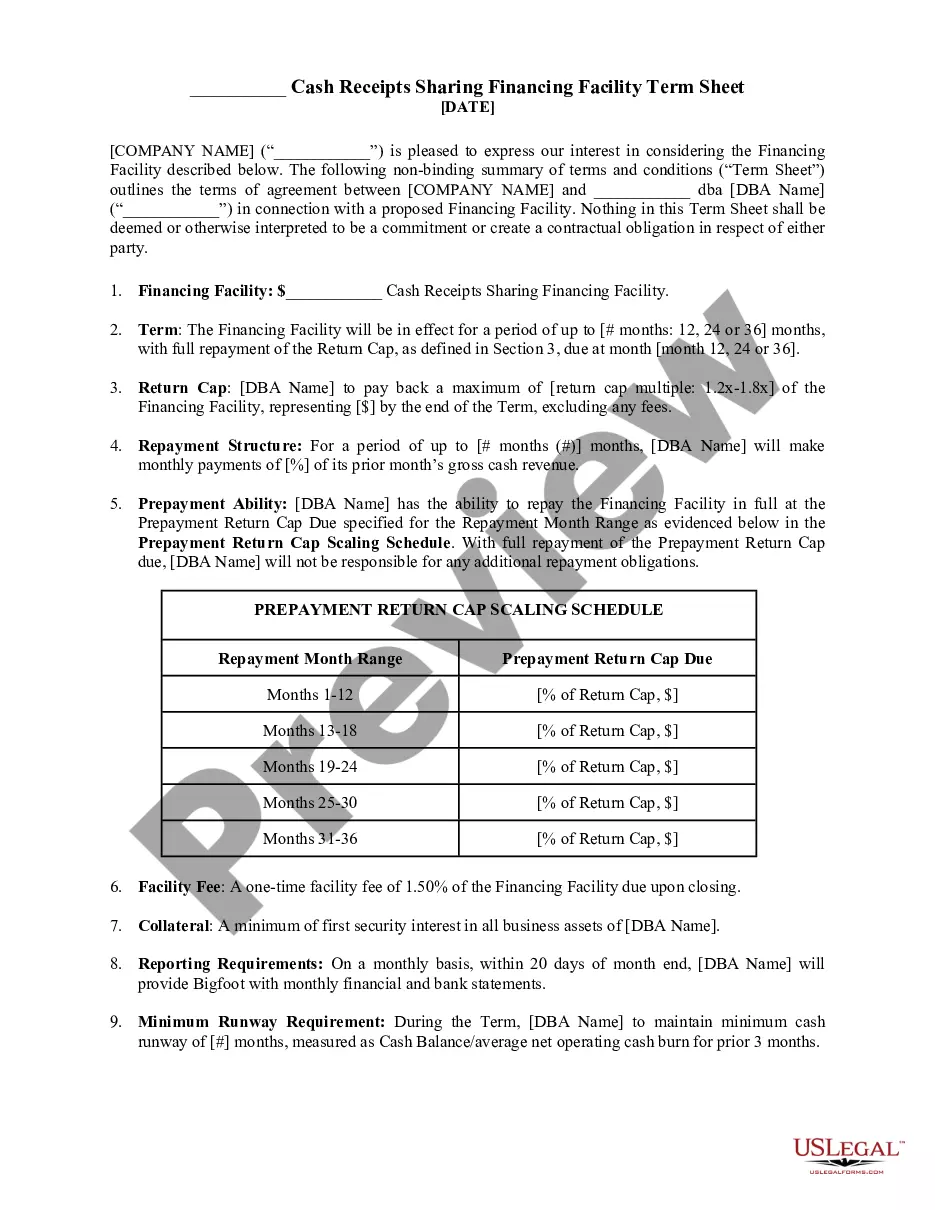
Description
Form popularity
FAQ
This legal agreement governs the disclosure of confidential or proprietary information as it relates to a clinical research study. Sponsors will often require an executed CDA in order to release the study protocol.
Definition: An NDA allows a party to communicate nonpublic information to another. A CDA allows multiple parties to handle information with utmost secrecy.
Just like any other contracts, an employee NDA is a legally binding document. As such, breaking an NDA would have adverse legal consequences against the employee.
A confidentiality agreement should include the names and addresses of the parties to the contract. Consider also including: Reason for the agreement: Explain why you're sharing this information. The information disclosed: Be specific about the subject matter and what exactly is included in the agreement.
What is a confidentiality agreement? Confidentiality agreements protect parties entering into business relationships or transactions that require the exchange of sensitive, private information otherwise inaccessible to third parties. Confidential information is the heart of any confidentiality agreement.
Confidentiality: Prevention of disclosure, to other than authorized individuals, of a sponsor's proprietary information or of a subject's identity. Direct Access: Permission to examine, analyze, verify, and reproduce any records and reports that are important to evaluation of a clinical trial.
In academic research confidentiality agreements are frequently used between researchers and members of their research team (e.g. translators, transcribers, student employees) who may be privy to confidential and/or sensitive information as part of their role in a research project.
Typically, a legal professional writing the NDA will complete these steps: Step 1 - Describe the scope. Which information is considered confidential? ... Step 2 - Detail party obligations. Step 3 - Note potential exclusions. Step 4 - Set the term. Step 5 - Spell out consequences.
I, (name of recipient) (the “Recipient”), agree as follows: To keep all the research information shared with me confidential by not discussing or sharing the research information in any form or format (e.g. disks, tapes, transcripts) with anyone other than the Principal Investigator(s);