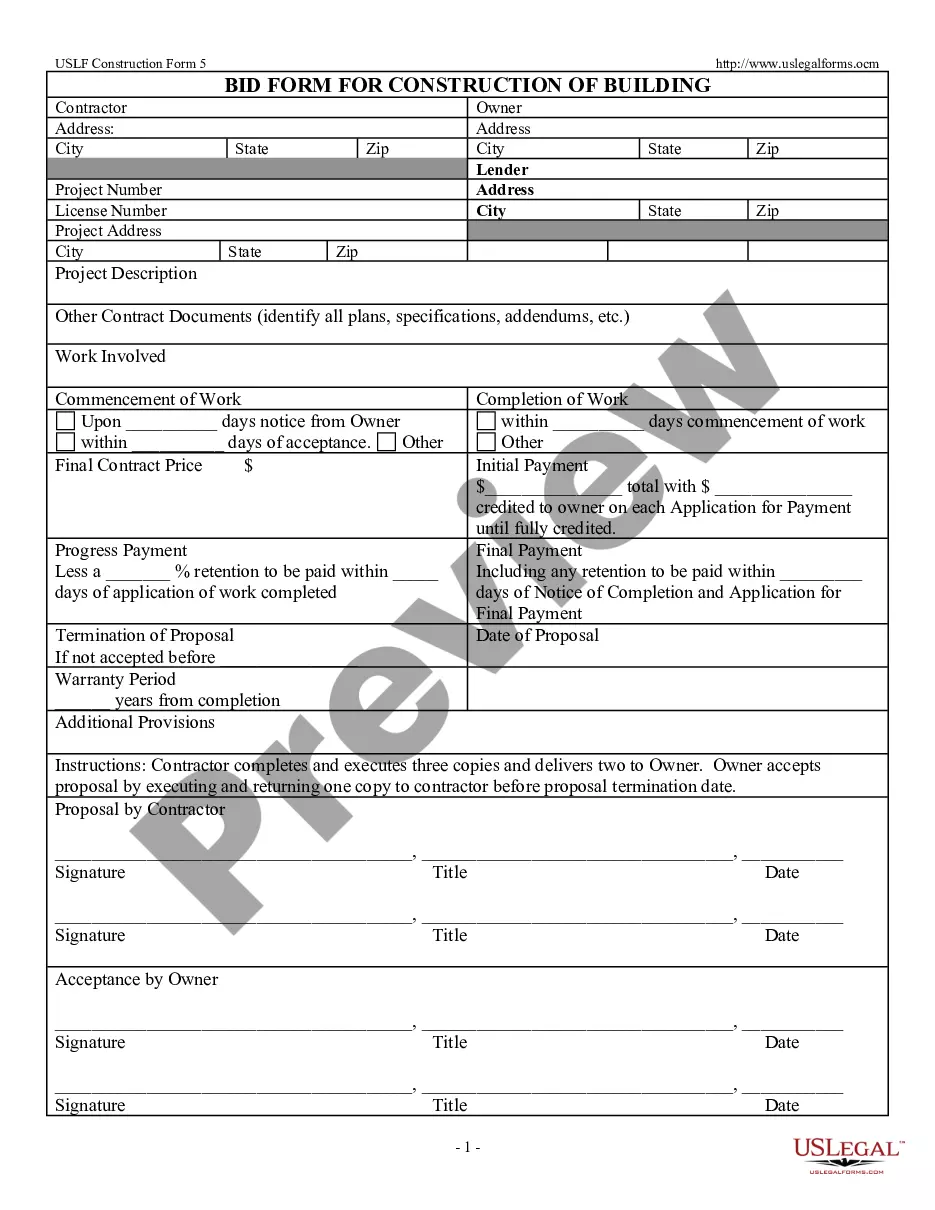
Consent Release Information Form Psychologist In Chicago
Description
Form popularity
FAQ
Instructions for Developing an Informed Consent Document General Information. Describe the purpose(s) of this research study in lay terms. Purpose of the Study. Procedures. Risks. Benefits. Compensation, Costs and Reimbursement. Withdrawal or Termination from Study. Confidentiality.
At a minimum, a well-designed informed consent form will address the following information: Risks and benefits of treatment. Fees and payment policies. Confidentiality and its limits. Contact information and communication. Social media policy and general boundaries. Emergency procedures.
The five essential elements of the informed consent process are Voluntary, Information, Understanding, Capacity, and Consent. Voluntary: Informed consent should be given voluntarily without any coercion or pressure.
Consent to treatment means a person must give permission before they receive any type of medical treatment, test or examination. This must be done on the basis of an explanation by a clinician. Consent from a patient is needed regardless of the procedure, whether it's a physical examination or something else.
Normally, psychotherapy is not a one-time intervention, but continues over a period of time or may be intermittent. Similarly, informed consent is not simply obtained at one point in time and never thought of again. Ongoing consent is implied by the continuing attendance of a client at therapy sessions.
The entire informed consent process involves giving a subject adequate information concerning the study, providing adequate opportunity for the subject to consider all options, responding to the subject's questions, ensuring that the subject has comprehended this information, obtaining the subject's voluntary agreement ...
Instructions for Developing an Informed Consent Document General Information. Describe the purpose(s) of this research study in lay terms. Purpose of the Study. Procedures. Risks. Benefits. Compensation, Costs and Reimbursement. Withdrawal or Termination from Study. Confidentiality.